Eradication of Helicobacter pylori with First-Line Treatment at a High-Complexity Hospital in Southwestern Colombia
DOI:
https://doi.org/10.22516/25007440.1229Keywords:
Helicobacter pylori, Eradication, Breath test, Microbial resistance, Treatment adherenceAbstract
Introduction and Objectives: Confirming the eradication of Helicobacter pylori is essential due to increasing antimicrobial resistance to various treatment regimens. The primary objective of this study is to determine the eradication rate of H. pylori using the carbon-14 (14C) urea breath test.
Materials and Methods: A cross-sectional study with an analytical component was conducted by retrospectively reviewing medical records of patients who received eradication treatment for H. pylori and subsequent confirmation of eradication through the 14C urea breath test. The study was carried out at Hospital Universitario Fundación Valle del Lili between January 2019 and June 2022.
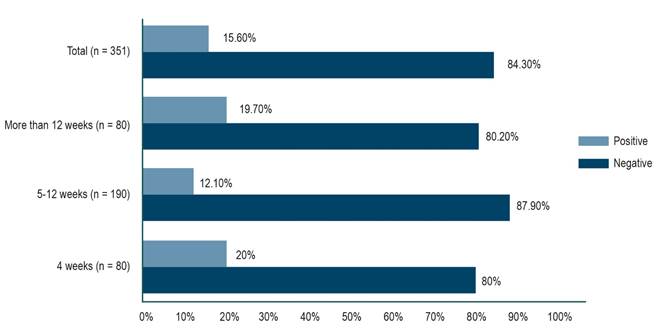
Results: A total of 360 patients met the inclusion criteria. Women represented 66.9% of the sample, with a median age of 51 years (interquartile range [IQR]: 39–61). A negative 14C urea breath test result was obtained in 84.4% of cases, with a median interval of eight weeks (IQR: 5–12) between the end of treatment and test performance. Almost all patients received 14 days of treatment.
Conclusions: An acceptable eradication rate was observed in our setting, even with the standard triple therapy, which remains the most commonly used regimen. Antimicrobial susceptibility studies are needed to guide treatments based on local epidemiology.
Downloads
References
Savoldi A, Carrara E, Graham DY, Conti M, Tacconelli E. Prevalence of Antibiotic Resistance in Helicobacter pylori: A Systematic Review and Meta-analysis in World Health Organization Regions. Gastroenterology. 2018;155(5):1372-1382.e17. https://doi.org/10.1053/j.gastro.2018.07.007
Sharara AI. Confirmatory Testing for Eradication of Helicobacter pylori: Challenges and Opportunities. Clin Gastroenterol Hepatol. 2021;19(2):232-234. https://doi.org/10.1016/j.cgh.2020.05.051
Li Y, Choi H, Leung K, Jiang F, Graham DY, Leung WK. Global prevalence of Helicobacter pylori infection between 1980 and 2022: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2023;8(6):553-564. https://doi.org/10.1016/S2468-1253(23)00070-5
Bravo LE, Matta AJ, Zambrano DC. Inmunotipificación de linfocitos en mucosa gástrica de pacientes infectados por H. pylori en dos regiones geográficas con contraste en el riesgo de desarrollar cáncer gástrico. Andes Pediatr. 2022;93(3):400-409. http://doi.org/10.32641/andespediatr.v93i3.3667
Bravo LE, Cortés A, Carrascal E, Jaramillo R, García LS, Bravo PE, et al. Helicobacter pylori: patología y prevalencia en biopsias gástricas en Colombia. Colombia Médica. 2003;34(3):124-131.
Correa S, Cardona AF, Correa T, García HI, Estrada S. Prevalencia de Helicobacter pylori y características histopatológicas en biopsias gástricas de pacientes con síntomas dispépticos en un centro de referencia de Medellín. Rev Colomb Gastroenterol. 2016;31(1):9-15. https://doi.org/10.22516/25007440.67
Sepúlveda M, Maldonado C, Bravo J, et al. Prevalence of Helicobacter pylori in patients undergoing upper digestive tract endoscopy at a referral hospital in Cali, Colombia, in 2020. Rev Colomb Gastroenterol. 2020;37(4):355-361. https://doi.org/10.22516/25007440.868
Chey WD, Leontiadis GI, Howden CW, Moss SF. Clinical Guideline: Treatment of Helicobacter pylori Infection. Am J Gastroenterol. 2017;112(2):212-239. https://doi.org/10.1038/ajg.2016.563
Sugano K, Tack J, Kuipers EJ, Graham DY, El-Omar EM, Miura S, et al. Kyoto global consensus report on Helicobacter pylori gastritis. Gut. 2015;64(9):1353-67. https://doi.org/10.1136/gutjnl-2015-309252
Kato M, Ota H, Okuda M, Kikuchi S, Satoh K, Shimoyama T, et al. Guidelines for the management of Helicobacter pylori infection in Japan: 2016 Revised Edition. Helicobacter 2019;24(4):e12597. https://doi.org/10.1111/hel.12597
Malfertheiner P, Megraud F, Rokkas T, Gisbert JP, Liou JM, Schulz C, et al. Management of Helicobacter pylori infection: the Maastricht VI/Florence consensus report. Gut. 2022:gutjnl-2022-327745. https://dx.doi.org/10.1136/gutjnl-2022-327745
Otero RW, Trespalacios RA, Otero PL, Vallejo MT, Torres Amaya M, Pardo R, et al. Guía de práctica clínica para el diagnóstico y tratamiento de la infección por Helicobacter pylori en adultos. Rev Colomb Gastroenterol. 2015;30(1):17-33.
Mahachai V, Vilaichone RK, Pittayanon R, Rojborwonwitaya J, Leelakusolvong S, Maneerattanaporn M, et al. Helicobacter pylori management in ASEAN: The Bangkok consensus report. J Gastroenterol Hepatol 2018;33(1):37-56. https://doi.org/10.1111/jgh.13911
Fallone CA, Chiba N, van Zanten SV, Fischbach L, Gisbert JP, Hunt RH, et al. The Toronto Consensus for the Treatment of Helicobacter pylori Infection in Adults. Gastroenterology. 2016;151(1):51-69.e14. https://doi.org/10.1053/j.gastro.2016.04.006
Liou JM, Malfertheiner P, Lee YC, Sheu BS, Sugano K, Cheng HC, et al. Screening and eradication of Helicobacter pylori for gastric cancer prevention: the Taipei global consensus. Gut. 2020;69(12):2093-2112. https://doi.org/10.1136/gutjnl-2020-322368
Roszczenko-Jasińska P, Wojtyś M, Jagusztyn-Krynicka E. Helicobacter pylori treatment in the post-antibiotics era—searching for new drug targets. Appl Microbiol Biotechnol 2020;104(23):9891-9905. https://doi.org/10.1007/s00253-020-10945-w
Wang X, Zhang S, Chua EG, He Y, Li X, Liu A, et al. A re-testing range is recommended for 13C- and 14C-urea breath tests for Helicobacter pylori infection in China. Gut Pathog. 2021;13(1):38. https://doi.org/10.1186/s13099-021-00435-3
Miftahussurur M, Windia A, Fahrial Syam A, Nusi IA, Alfaray RI, Fauzia KA, et al. Diagnostic Value of 14C Urea Breath Test for Helicobacter pylori Detection Compared by Histopathology in Indonesian Dyspeptic Patients. Clin Exp Gastroenterol. 2021;14:291-296. https://doi.org/10.2147/CEG.S306626
Vaira D, Vakil N. Blood, urine, stool, breath, money, and Helicobacter pylori. Gut. 2001;48(3):287-28. https://doi.org/10.1136/gut.48.3.287
Cutler AF, Havstad S, Ma CK, Blaser MJ, Perez-Perez GI, Schubert TT. Accuracy of invasive and noninvasive tests to diagnose Helicobacter pylori infection. Gastroenterology. 1995;109(1):136-41. https://doi.org/10.1016/0016-5085(95)90278-3
Best LM, Takwoingi Y, Siddique S, Selladurai A, Gandhi A, Low B, et al. Non-invasive diagnostic tests for Helicobacter pylori infection. Cochrane Database Syst Rev. 2018;3(3):CD012080. https://doi.org/10.1002/14651858.CD012080.pub2
Gisbert JP, Pajares JM. Review article: 13C-urea breath test in the diagnosis of Helicobacter pylori infection -- a critical review. Aliment Pharmacol Ther. 2004;20(10):1001-17. https://doi.org/10.1111/j.1365-2036.2004.02203.x
Katelaris P, Hunt R, Bazzoli F, Cohen H, Fock KM, Gemilyan M, et al. Helicobacter pylori World Gastroenterology Organization Global Guideline. J Clin Gastroenterol. 2023;57(2):111-126. https://doi.org/10.1097/MCG.0000000000001719
Graham DY, Lee YC, Wu MS. Rational Helicobacter pylori therapy: evidence-based medicine rather than medicine-based evidence. Clin Gastroenterol Hepatol 2014;12(2):177-186.e173. https://doi.org/10.1016/j.cgh.2013.05.028
Matsumoto H, Shiotani A, Graham D. Current and Future Treatment of Helicobacter pylori Infections. Adv Exp Med Biol. 2019;1149:211-225. https://doi.org/10.1007/5584_2019_367
Trespalacios AA, Otero W, Mercado M. Resistencia de Helicobacter pylori a metronidazol, claritromicina y amoxicilina en pacientes colombianos 2010. Rev Colomb Gastroenterol. 2010;25(1):31-38.
Yepes CA, Rodríguez Varón A, Ruiz Morales A, Ariza B. Resistencia antibiótica del Helicobacter pylori en el Hospital Universitario San Ignacio de Bogotá. Acta Médica Colombiana. 2008;33(1):11-34.
Figueroa M, Cortés A, Pazos Álvaro, Bravo LE. Sensibilidad in vitro a amoxicilina y claritromicina de Helicobacter pylori obtenido de biopsias gástricas de pacientes en zona de bajo riesgo para cáncer gástrico. Biomédica. 2012;32(1):32-4. https://doi.org/10.7705/biomedica.v32i1.454
Álvarez A, Moncayo JI, Santacruz JJ, Corredor LF, Reinosa E, Martínez JW, et al. Antimicrobial susceptibility of Helicobacter pylori strains isolated in Colombia. Rev Med Chil. 2009;137(10):1309-1314.
Trespalacios AA, Otero W, Caminos JE, Mercado MM, Avila J, Rosero LE, et al. Phenotypic and genotypic analysis of clarithromycin-resistant Helicobacter pylori from Bogotá D.C., Colombia. J Microbiol. 2013;51(4):448-52. https://doi.org/10.1007/s12275-013-2465-6
Camargo MC, García A, Riquelme A, Otero W, Camargo CA, Hernandez-García T, et al. The problem of Helicobacter pylori resistance to antibiotics: a systematic review in Latin America. Am J Gastroenterol. 2014;109(4):485-495. https://doi.org/10.1038/ajg.2014.24
Martínez JD, Henao SC, Lizarazo JI. Resistencia antibiótica del Helicobacter pylori en América Latina y el Caribe. Rev Col Gastroenterol. 2014;29(3):218-227.
Acosta CP, Quiroga AJ, Sierra CH, Trespalacios AA. Frecuencia de mutaciones de la nitrorreductasa RdxA de Helicobacter pylori para la activación del metronidazol en una población del departamento del Cauca, Colombia. Biomédica. 2017;37(2):191-9. https://doi.org/10.7705/biomedica.v37i2.3007
Trespalacios-Rangel A, Otero W, Arévalo-Galvis A, Poutou-Piñales RA, Rimbara E, Graham DY. Surveillance of Levofloxacin Resistance in Helicobacter pylori Isolates in Bogotá-Colombia (2009-2014). PloS One. 2016;11(7):e0160007. https://doi.org/10.1371/journal.pone.0160007
Chen Y, Yuan H, Ye H, Shi Z, Deng X, Zhang X, et al. Application of a semi-automatic, intensive follow-up for improving efficacy and adherence of Helicobacter pylori eradication therapy: A randomized controlled trial. Microbiologyopen. 2021;10(1):e1172. https://doi.org/10.1002/mbo3.1172
Salazar D, Mejía T, Moncayo J, Guaca Gonzales YM, Arturo BL, Pacheco López R, et al. Frecuencia, características clínicas y sociodemográficas relacionadas al abandono del tratamiento en un grupo de pacientes con diagnóstico y tratamiento de H. pylori en centros especializados de Pereira y Manizales. Revista De Investigación De La Universidad Norbert Wiener. 2022;11(2):a0010. https://doi.org/10.37768/unw.rinv.11.02.a0010
Malfertheiner P, Megraud F, O’Morain CA, Gisbert JP, Kuipers EJ, Axon AT, et al. Management of Helicobacter pylori infection-the Maastricht V/Florence Consensus Report. Gut. 2017;66(1):6-30. https://doi.org/10.1136/gutjnl-2016-312288
Vargas Cárdenas G, Balvin Yanes L, Chaiña Meza JM, Llanos Tejada F. Adherencia terapéutica al tratamiento de erradicación de Helicobacter pylori y sus factores asociados en un hospital público de Perú. Rev. gastroenterol. Perú. 2020;40(3):224-229.
Smith S, Jolaiya T, Fowora M, Palamides P, Ngoka F, Bamidele M, et al. Clinical and Socio- Demographic Risk Factors for Acquisition of Helicobacter pylori Infection in Nigeria. Asian Pac J Cancer Prev. 2018;19(7):1851-1857. https://doi:10.22034/APJCP.2018.19.7.1851
Sánchez Ceballos F, Taxonera Samsó C, García Alonso M, Alba López C, Sainz de los Terreros Soler L, Díaz-Rubio M. Prevalencia de la infección por Helicobacter pylori en población sana en la Comunidad de Madrid. Rev Esp Enferm Dig. 2007;99(9):497-501.
Ren S, Cai P, Liu Y, Wang T, Zhang Y, Li Q, et al. Prevalence of Helicobacter pylori infection in China: A systematic review and meta-analysis. J Gastroenterol Hepatol. 2022;37(3):464-470. https://doi.org/10.1111/jgh.15751
Laine L, Estrada R, Trujillo M, Knigge K, Fennerty MB. Effect of proton-pump inhibitor therapy on diagnostic testing for Helicobacter pylori. Ann Intern Med. 1998 Oct 1;129(7):547-50. https://doi.org/10.7326/0003-4819-129-7-199810010-00007
Capurso G, Carnuccio A, Lahner E, Panzuto F, Baccini F, Delle Fave G, et al. Corpus-predominant gastritis as a risk factor for false-negative 13C-urea breath test results. Aliment Pharmacol Ther. 2006;24(10):1453-60. https://10.1111/j.1365-2036.2006.03143.x
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2024 Revista colombiana de Gastroenterología

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Aquellos autores/as que tengan publicaciones con esta revista, aceptan los términos siguientes:
Los autores/as ceden sus derechos de autor y garantizarán a la revista el derecho de primera publicación de su obra, el cuál estará simultáneamente sujeto a la Licencia de reconocimiento de Creative Commons que permite a terceros compartir la obra siempre que se indique su autor y su primera publicación en esta revista.
Los contenidos están protegidos bajo una licencia de Creative Commons Reconocimiento-NoComercial-SinObraDerivada 4.0 Internacional.