Rol del farmacéutico en la atención integral de pacientes con hepatitis C: revisión sistemática
DOI:
https://doi.org/10.22516/25007440.510Palabras clave:
Servicios Farmacéuticos, Manejo de Atención al Paciente, Hepatitis C, Antivirales, Atención Integral de SaludResumen
Objetivo: Identificar y caracterizar las acciones/intervenciones realizadas desde los servicios farmacéuticos en el abordaje de pacientes con Hepatitis C y proponer una vía clínica para la gestión de la enfermedad que incluya la participación del farmacéutico.
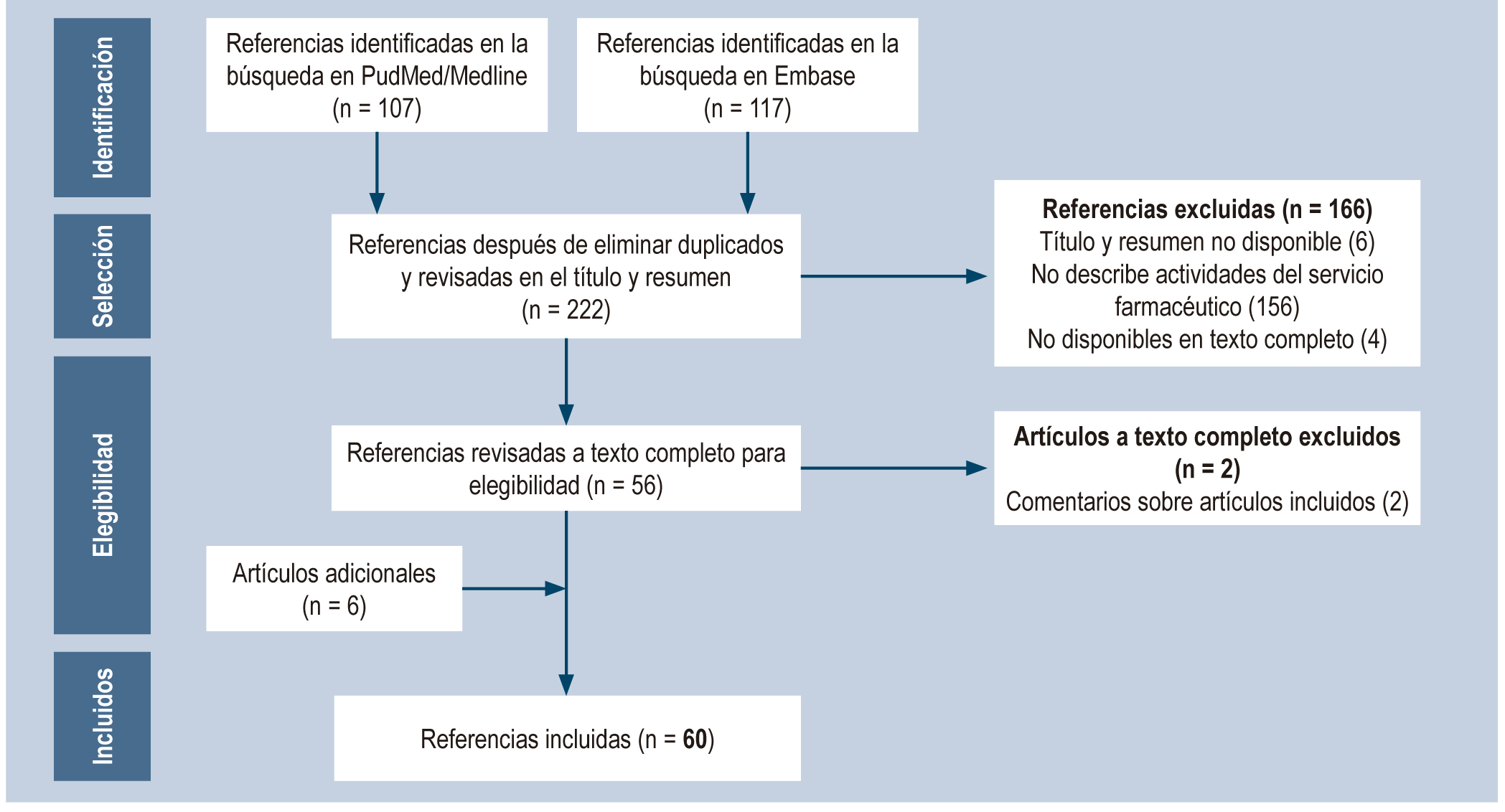
Método: Revisión sistemática en PubMed y EMBASE empleando los términos "Hepatitis C", "Pharmaceutical Services", "Community Pharmacy Services", y "Pharmacies"; artículos publicados hasta el 31 de marzo de 2019, cuyo tema central fueran las actividades realizadas por los servicios farmacéuticos en la atención a pacientes con Hepatitis C. Se recopiló información sobre: actividad realizada, responsable, si la intervención era individual o colectiva y el entorno de aplicación. Las actividades se agruparon en: promoción y prevención, gestión administrativa, atención farmacéutica, investigación y otros servicios de apoyo. De acuerdo a esto, se propuso una vía clínica para el manejo de la Hepatitis C con participación del farmacéutico.
Resultados: se incluyeron 60 artículos, principalmente de estudios descriptivos. La mayoria de las publicaciones reportaron intervenciones realizadas en Estados Unidos y España. Se identificó la participación del personal farmacéutico en cada una de las etapas del proceso de atención, incluyendo: provisión de servicios de reducción del daño, tamizaje del Virus de la Hepatitis C, vinculación de los pacientes al tratamiento, prescripción de medicamentos y órdenes de laboratorio.
Conclusiones: Se identifican y caracterizan las acciones/intervenciones realizadas desde el servicio farmacéutico para el manejo de la Hepatitis C y se propone una vía clínica en la que se integran los servicios profesionales farmacéuticos a las demás actividades de la atención del paciente.
Descargas
Referencias bibliográficas
Tran AN, Sachdev R, Fricker ZP, Leber M, Zahorian T, Shah B, Nunes DP, Long MT. Intensive Pharmacy Care Improves Outcomes of Hepatitis C Treatment in a Vulnerable Patient Population at a Safety-Net Hospital. Dig Dis Sci. 2018;63(12):3241-9.
https://doi.org/10.1007/s10620-018-5231-0
Andres J, Lott S, Qureshi K. Eight-Week Outcomes of Ledipasvir/Sofosbuvir in Noncirrhotic Treatment-Naive Patients with Hepatitis C: Analysis of Pharmacy-Based Data. J Manag Care Spec Pharm. 2018;24(1):23-8.
https://doi.org/10.18553/jmcp.2018.24.1.23
Gauthier TP, Moreira E, Chan C, Cabrera A, Toro M, Carrasquillo MZ, Corentin M, Sherman EM. Pharmacist engagement within a hepatitis C ambulatory care clinic in the era of a treatment revolution. J Am Pharm Assoc JAPhA. 2016;56(6):670-6.
https://doi.org/10.1016/j.japh.2016.06.013
Galewitz P. VA Shifts To Clinical Pharmacists To Help Ease Patients’ Long Waits [Internet]. Kaiser Health News; 2016 [citado el 28 de abril de 2018]. Disponible en: https://khn.org/news/va-treats-patients-impatience-with-clinical-pharmacists/
You A, Kawamoto J, Smith JP. A pharmacist-managed telemedicine clinic for hepatitis C care: a descriptive analysis. J Telemed Telecare. 2014;20(2):99-101.
https://doi.org/10.1177/1357633X13519043
Dunn EE, Vranek K, Hynicka LM, Gripshover J, Potosky D, Mattingly TJ. Evaluating a Collaborative Approach to Improve Prior Authorization Efficiency in the Treatment of Hepatitis C Virus. Qual Manag Health Care. 2017;26(3):136-9.
https://doi.org/10.1097/QMH.0000000000000137
Calderon Y, Cowan E, Schramm C, Stern S, Brusalis C, Iscoe M, Rahman S, Verma R, Leider J. HCV and HBV testing acceptability and knowledge among urban emergency department patients and pharmacy clients. Prev Med. 2014;61:29-33.
https://doi.org/10.1016/j.ypmed.2013.12.026
Mohammad RA, Bulloch MN, Chan J, Deming P, Love B, Smith L, Dong BJ, GI Liver Nutrition and Infectious Diseases Practice and Research Networks of the American College of Clinical Pharmacy. Provision of clinical pharmacist services for individuals with chronic hepatitis C viral infection: Joint Opinion of the GI/Liver/Nutrition and Infectious Diseases Practice and Research Networks of the American College of Clinical Pharmacy. Pharmacotherapy. 2014;34(12):1341-54.
https://doi.org/10.1002/phar.1512
Dong BJ, Lopez M, Cocohoba J. Pharmacists performing hepatitis C antibody point-of-care screening in a community pharmacy: A pilot project. J Am Pharm Assoc JAPhA. 2017;57(4):510-515.e2.
https://doi.org/10.1016/j.japh.2017.04.463
Radley A, Tait J, Dillon JF. DOT-C: A cluster randomised feasibility trial evaluating directly observed anti-HCV therapy in a population receiving opioid substitute therapy from community pharmacy. Int J Drug Policy. 2017;47:126-36.
https://doi.org/10.1016/j.drugpo.2017.05.042
Iizuka T, Eguchi Y, Akase T, Ishizuka H, Yoshiyama Y. Primary Care Enlightenment to Local Inhabitants -Cooperation of Medical Institution and Community Pharmacy in Treatment of Chronic Hepatitis C. Yakugaku Zasshi. 2010;130(12):1633-9.
https://doi.org/10.1248/yakushi.130.1633
Cooper EN, Dodson C, Stopka TJ, Riley ED, Garfein RS, Bluthenthal RN. Pharmacy participation in non-prescription syringe sales in Los Angeles and San Francisco counties, 2007. J Urban Health Bull N Y Acad Med. 2010;87(4):543-52.
https://doi.org/10.1007/s11524-010-9483-z
Garfein RS, Stopka TJ, Pavlinac PB, Ross A, Haye BK, Riley ED, Bluthenthal RN. Three years after legalization of nonprescription pharmacy syringe sales in California: where are we now? J Urban Health Bull N Y Acad Med. 2010;87(4):576-85.
https://doi.org/10.1007/s11524-010-9463-3
Stancliff S, Agins B, Rich JD, Burris S. Syringe access for the prevention of blood borne infections among injection drug users. BMC Public Health. 2003;3:37.
https://doi.org/10.1186/1471-2458-3-37
Yang Y, Latkin CA, Luan R, Yang C. A cross-sectional study of the feasibility of pharmacy-delivered harm reduction services among people who inject drugs in Xichang, China. BMC Public Health. 2015;15:885.
https://doi.org/10.1186/s12889-015-2236-x
Oramasionwu CU, Johnson TL, Zule WA, Carda-Auten J, Golin CE. Using Pharmacies in a Structural Intervention to Distribute Low Dead Space Syringes to Reduce HIV and HCV Transmission in People Who Inject Drugs. Am J Public Health. 2015;105(6):1066-71.
https://doi.org/10.2105/AJPH.2015.302581
Isho NY, Kachlic MD, Marcelo JC, Martin MT. Pharmacist-initiated hepatitis C virus screening in a community pharmacy to increase awareness and link to care at the medical center. J Am Pharm Assoc JAPhA. 2017;57(3S):S259-64. https://doi.org/10.1016/j.japh.2017.03.006
Pharmacy hepatitis project results positive. The Pharmaceutical Journal. 15 de enero de 2010; News & analysis: 35.
Showande S, Olaifa A. Community pharmacists ‘ involvement in the ordering and interpretation of laboratory tests. Int J Pharm Sci Res. 2013;4(3):988-96.
Briggs AL. Pharmacists’ increasing involvement in hepatitis C management and prevention. J Am Pharm Assoc JAPhA. 2018;58(1):5-6.
https://doi.org/10.1016/j.japh.2017.12.008
D’Angelo RG, Klepser M, Woodfield R, Patel H. Hepatitis C Virus Screening: A Review of the OraQuick Hepatitis C Virus Rapid Antibody Test. J Pharm Technol. 2015;31(1):13-9.
https://doi.org/10.1177/8755122514548228
Cohen SM, Kwasny MJ, Ahn J. Use of specialty care versus standard retail pharmacies for treatment of hepatitis C. Ann Pharmacother. 2009;43(2):202-9.
https://doi.org/10.1345/aph.1L227
Martin MT, Faber DM. Patient satisfaction with the clinical pharmacist and prescribers during hepatitis C virus management. J Clin Pharm Ther. 2016;41(6):645-9.
https://doi.org/10.1111/jcpt.12436
Pham TT, Keast SL, Farmer KC, Thompson DM, Rathbun RC, Nesser NJ, Holderread BP, Skrepnek GH. Sustained Virologic Response and Costs Associated with Direct-Acting Antivirals for Chronic Hepatitis C Infection in Oklahoma Medicaid. J Manag Care Spec Pharm. 2018;24(7):664-76.
https://doi.org/10.18553/jmcp.2018.24.7.664
Martin MT, Telebak E, Taylor PA, Volozhina O. Development of a specialty medication prior-authorization service at an urban academic medical center. Am J Health Syst Pharm. 2016;73(15):1174-9.
https://doi.org/10.2146/ajhp160059
Vu TM, Toribio W, Riazi F, Ciprian G, Gibbs N, Giardina M, Camacho JA, Parrella K, Cambe J, Amory C, Chasan R, Sigel KM, Weiss JJ. Increasing Access to Hepatitis C Virus Medications: A Program Model Using Patient Navigators and Specialty Pharmacy to Obtain Prior Authorization Approval. J Manag Care Spec Pharm. 2018;24(4):329-33. https://doi.org/10.18553/jmcp.2018.24.4.329
Grischeau M, Zenner J. Optimizing workflow at a multidisciplinary clinic for management of hepatitis C virus infection. Am J Health-Syst Pharm AJHP Off J Am Soc Health-Syst Pharm. 2012;69(24):2131-3.
https://doi.org/10.2146/ajhp120264
Ribed A, Rodriguez-González C, Collado-Borrell R, Camino S, Elena L, Alvaro G, Ibañez-García S, Tovar-Pozo M, Herranz-Alonso AM, Sanjurjo-Sáez M. CP-102 Pharmaceutical care monitoring of hepatitis C outpatients: Guaranteeing safety and efficiency. Eur J Hosp Pharm. 2016;23(Suppl 1):A44.3-A45.
https://doi.org/10.1136/ejhpharm-2016-000875.102
Márquez-Peiró JF, Pérez-Peiró C, Carmena-Carmena J, Jiménez-Torres NV. Identifying improvement opportunities in the management of hepatitis C. Farm Hosp. 2006;30(3):154-60.
https://doi.org/10.1016/S1130-6343(06)73966-6
Henderson RR, Visaria J, Bridges GG, Dorholt M, Levin RJ, Frazee SG. Impact of specialty pharmacy on telaprevir-containing 3-drug hepatitis C regimen persistence. J Manag Care Spec Pharm. 2014;20(12):1227-34.
https://doi.org/10.18553/jmcp.2014.20.12.1227
Foisy MM, Akai PS. Pharmaceutical care for HIV patients on directly observed therapy. Ann Pharmacother. 2004;38(4):550-6.
https://doi.org/10.1345/aph.1D444
Lavitas P, Tesell M, Hydery T, Greenwood BC, Price M, Lenz K, Jeffrey P. Overview of Comprehensive Hepatitis C Virus Medication Management in a State Medicaid Program. J Manag Care Spec Pharm. 2016;22(10):1161-6. https://doi.org/10.18553/jmcp.2016.22.10.1161
Chamorro-de-Vega E, Rodriguez-Gonzalez CG, Gimenez-Manzorro A, de Lorenzo-Pinto A, Iglesias-Peinado I, Herranz A, Sanjurjo M; GRUviC Study Group. Improving pharmacotherapy outcomes in patients with hepatitis C virus infection treated with direct-acting antivirals: The GRUviC project. Int J Clin Pract. 2017;71(8):e12988. https://doi.org/10.1111/ijcp.12988
Kolor B. Patient education and treatment strategies implemented at a pharmacist-managed hepatitis C virus clinic. Pharmacotherapy. 2005;25(9):1230-41.
https://doi.org/10.1592/phco.2005.25.9.1230
Zaepfel M, Cristofaro L, Trawinski A, McCarthy K, Rightmier E, Khadem T. Evaluation of a Hepatitis C Patient Management Program at a University Specialty Pharmacy. Ann Pharmacother. 2017;51(4):307-14.
https://doi.org/10.1177/1060028016683495
Gomes LO, Teixeira MR, Rosa JAD, Foppa AA, Rover MRM, Farias MR. The benefits of a public pharmacist service in chronic hepattis C treatment: The real-life results of sofosbuvir-based therapy. Res Social Adm Pharm. 2020;16(1):48-53.
https://doi.org/10.1016/j.sapharm.2019.02.008
Collado-Borrell R, Lallana-Sainz E, Gimenez-Manzorro A, Ribed-Sanchez A, Lorenzo-Pinto AD, Chamorro E, Romero-Jimenez R, Tovar-Pozo M, Herranz-Alonso A, Sanjurjo-Saez M. PS-020 Drug interactions of new direct acting antiviral agents detected in an intensive pharmaceutical care programme of hepatits C patients. Eur J Hosp Pharm. 2016;23(Suppl 1):A222-3.
https://doi.org/10.1136/ejhpharm-2016-000875.505
Masip M, Tuneu L, Pagès N, Torras X, Gallego A, Guardiola JM, Faus MJ, Mangues MA. Prevalence and detection of neuropsychiatric adverse effects during hepatitis C treatment. Int J Clin Pharm. 2015;37(6):1143-51. https://doi.org/10.1007/s11096-015-0177-1
Robustillo M de las A, Almeida-Gonzalez CV, Morillo-Verdugo R. Relación entre la complejidad farmacoterapéutica y la satisfacción del paciente con el tratamiento. Farm Hosp. 2017;(4):470-8.
Cañamares Orbis I, Saez de la Fuente J, Izquierdo García E, Esteban Alba C, Such Díaz A, Escobar Rodriguez I. Experiencia autorreferida en pacientes tratados con antivirales directos frente al virus de la. Farm Hosp. 2016;(6):569-78.
Rodríguez-Camacho JM, Vela VV, Fernández MJH, Martín MVM, Bautista MJM, Gil LO. Monitoring of Pharmaceutical Care Hepatitis C program (2007-2011). Eur J Hosp Pharm. 2012;19(2):133.1-133.
https://doi.org/10.1136/ejhpharm-2012-000074.127
García-Pelayo M, Montilla E, Fuentes B. CP-079 Pharmaceutical Care for patients with hepatitis C treated with telaprevir. Role in the Regional Hospital. Eur J Hosp Pharm. 2014;21(Suppl 1):A32.3-A33.
https://doi.org/10.1136/ejhpharm-2013-000436.78
44th ESCP International Symposium on Clinical Pharmacy Medicines Information: Making Better Decisions: Lisbon, Portugal, 28-30 October 2015. Int J Clin Pharm. 2016;38(2):470-598.
https://doi.org/10.1007/s11096-015-0240-y
Yang S, Britt RB, Hashem MG, Brown JN. Outcomes of Pharmacy-Led Hepatitis C Direct-Acting Antiviral Utilization Management at a Veterans Affairs Medical Center. J Manag Care Spec Pharm. 2017;23(3):364-9. https://doi.org/10.18553/jmcp.2017.23.3.364
Margusino-Framiñán L, Cid-Silva P, Mena-de-Cea Á, Sanclaudio-Luhía AI, Castro-Castro JA, Vázquez-González G, Martín-Herranz I. Intelligent MONitoring System for antiviral pharmacotherapy in patients with chronic hepatitis C (SiMON-VC). Farm Hosp. 2017;41(n01):68-88. https://doi.org/10.7399/fh.2017.41.1.10590
Sebhatu P, Martin MT. Genotype 1 hepatitis C virus and the pharmacist’s role in treatment. Am J Health Syst Pharm. 2016;73(11):764-74.
https://doi.org/10.2146/ajhp150704
Belperio PS, Backus LI, Ross D, Neuhauser MM, Mole LA. A Population Approach to Disease Management: Hepatitis C Direct-Acting Antiviral Use in a Large Health Care System. J Manag Care Pharm. 2014;20(6):533-40.
https://doi.org/10.18553/jmcp.2014.20.6.533
Walters-Smith N, Marshall SM. Opportunities and considerations for pharmacist intervention in the management of the chronic hepatitis C patient. J Manag Care Pharm JMCP. 2009;15(5):417-9.
https://doi.org/10.18553/jmcp.2009.15.5.417
Marino EL, Alvarez-Rubio L, Miro S, Modamio P, Banos F, Lastra CF, Alberdi-Leniz A. Pharmacist intervention in treatment of patients with genotype 1 chronic hepatitis C. J Manag Care Pharm JMCP. 2009;15(2):147-50.
https://doi.org/10.18553/jmcp.2009.15.2.147
Yeste-Gómez I, Rodríguez-González CG, Giménez-Manzorro Á, Ais-Larisgoitia A, Sanjurjo-Saez M. Information leaflets for patients with hepatitis C receiving treatment with triple therapy. Eur J Hosp Pharm Sci Pract. 2013;20(1):13-9.
https://doi.org/10.1136/ejhpharm-2012-000176
Asavakarn S, Sirivatanauksorn Y, Promraj R, Ruenrom A, Limsrichamrern S, Kositamongkol P, Mahawithitwong P, Tovikkai C, Dumronggittigule W. Systematic Pharmaceutical Educational Approach to Enhance Drug Adherence in Liver Transplant Recipients. Transplant Proc. 2016;48(4):1202-7.
https://doi.org/10.1016/j.transproceed.2015.12.100
Smith JP, Dong MH, Kaunitz JD. Evaluation of a pharmacist-managed hepatitis C care clinic. Am J Health-Syst Pharm AJHP Off J Am Soc Health-Syst Pharm. 2007;64(6):632-6.
https://doi.org/10.2146/ajhp060153
Morillo-Verdugo R, Romero-Gómez M. Un nuevo escenario terapéutico en el tratamiento de la hepatitis crónica por virus C. Farm Hosp. 2012;36(6):466-8.
Marcoux RM, Simeone JC, Colavita M, Larrat EP. An Innovative Approach to Pharmacy Management in a State Correctional System. J Correct Health Care. 2012;18(1):53-61.
https://doi.org/10.1177/1078345811421732
Rodis JL, Kibbe P. Development of a hepatitis C support group. Am J Health Syst Pharm. 2006;63(17):1594-6. https://doi.org/10.2146/ajhp050481
Hussein M, Benner JS, Lee D, Sesti A-M, Battleman DS, Brock-Wood C. Propensity score matching in the evaluation of drug therapy management programs: an illustrative analysis of a program for patients with hepatitis C virus. Qual Manag Health Care. 2010;19(1):25-33.
https://doi.org/10.1097/QMH.0b013e3181ccbc7a
Veenstra DL, Higashi MK, Phillips KA. Assessing the cost-effectiveness of pharmacogenomics. AAPS PharmSci. 2000;2(3):80-90.
https://doi.org/10.1208/ps020329
Carnevale RC, de Godoi Rezende Costa Molino C, Visacri MB, Mazzola PG, Moriel P. Cost analysis of pharmaceutical care provided to HIV-infected patients: an ambispective controlled study. Daru J Fac Pharm Tehran Univ Med Sci. 2015;23(1):13.
https://doi.org/10.1186/s40199-014-0074-5
Menchen B, Folguera C, De rivas A, Saavedra V, Sanchez A. CP-149 Redesign of the management model and pharmaceutical care of patients with hepatitis C virus infection. Eur J Hosp Pharm. 2016;23(Suppl 1):A66.1-A66.
https://doi.org/10.1136/ejhpharm-2016-000875.149
Mehta BH, Rodis JL, Nahata MC, Bennett MS. Advancing patient care through innovative practice: The Clinical Partners Program. Am J Health Syst Pharm. 2005;62(23):2501-7.
https://doi.org/10.2146/ajhp050017
Ministerio de Salud y Protección Social, Instituto de Evaluación Tecnológica en Salud. Vía clínica para el tratamiento de hepatitis C. Bogotá, D.C: Ministerio de Salud y Protección Social; 2017.
Descargas
Publicado
Cómo citar
Número
Sección
Licencia
Aquellos autores/as que tengan publicaciones con esta revista, aceptan los términos siguientes:
Los autores/as ceden sus derechos de autor y garantizarán a la revista el derecho de primera publicación de su obra, el cuál estará simultáneamente sujeto a la Licencia de reconocimiento de Creative Commons que permite a terceros compartir la obra siempre que se indique su autor y su primera publicación en esta revista.
Los contenidos están protegidos bajo una licencia de Creative Commons Reconocimiento-NoComercial-SinObraDerivada 4.0 Internacional.

| Estadísticas de artículo | |
|---|---|
| Vistas de resúmenes | |
| Vistas de PDF | |
| Descargas de PDF | |
| Vistas de HTML | |
| Otras vistas | |
Datos de los fondos
-
Universidad de Antioquia
Números de la subvención CODI 2017-16258